 
She used several other names – magnesia, wismuth or the secretive ‘minera’ – even though German miners had earlier coined the word cobalt to describe the ores she studied. Wallich never used the word cobalt in her books. In spite of her detailed research Wallich, like the other chymists, was unable to create the philosophers’ stone. 
Wallich became a much sought-after chymist following the publication of her books. When heated and cooled, the compounds have colours ranging from rose, violet blue, sky blue and grass green. She is credited with discovering and reporting many thermochromic effects of cobalt compounds. Wallich’s books describe numerous chemical reactions in detail. Wallich’s goal was to produce the philosophers’ stone – a substance that could turn base metals such as lead and tin into silver and gold. Dorothea Juliana Wallich was interested in chymistry (a term that covers both alchemy and chemistry). Thirty years prior to Georg Brandt’s published account of separating cobalt from bismuth, a young German woman had already published three books about her research using ores that contained cobalt. When the temperature reaches the boiling point of any substance in the mixture, it will boil and can be collected as a pure gas, leaving the other components still in the liquid state.Chymical research and the philosophers’ stone Air can be separated into the component gases in the mixture by cooling it until it liquifies, the slowly raising the temperature of the liquid air. Molecules of carbon dioxide are formed from one carbon atom and two oxygen atoms chemically bonded together. Argon is made of individual argon atoms not joined to anything else. Molecules of oxygen are made from pairs of oxygen atoms chemically bonded together. It contains elements such as argon and oxygen, and compounds such as carbon dioxide. There are many physical means of separating a mixture: the right one to use depends on the physical states of the substances in the mixture. Unlike a compound, the different substances in a mixture can be separated from one another without breaking any chemical bonds. The substances in a mixture can be elements, compounds, or both. MixtureĪ mixture contains more than one chemical substance, and therefore is not pure. It is not possible to separate the atoms in a compound without breaking chemical bonds. All pure substances are either elements or compounds. CompoundĪ compound is a pure chemical substance, but it is made from more than one type of atom, chemically bonded together. 
The periodic table collects and organises all the known types of atom from which the elements are formed. For example, the element chlorine consists of pairs of chlorine atoms chemically bonded together, so its chemical formula is Cl 2. They may be joined to one another by chemical bonds, forming identical small groups of atoms called molecules. These atoms can be individual and separate, like in Noble Gases such as helium. ElementĪn element is a pure substance made up of only one type of atom. You should understand the difference between an atom and a molecule. 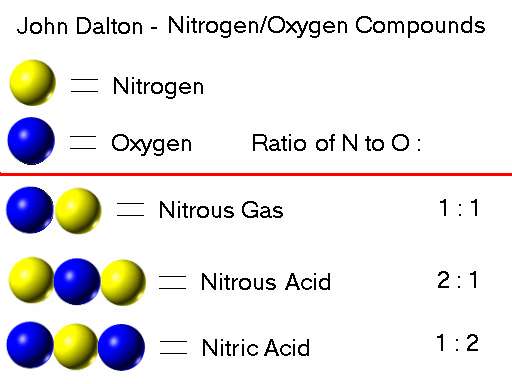
Prior learning: You should understand that everything is made of atoms, and understand the differences between different types of atom. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
